Project Overview
Ubiquitination is a post-translational modification process that controls the degradation, signalling and activity of many, if not all, cellular proteins. Ubiquitination needs to be finely tuned and is catalysed and controlled by several players: ubiquitin, the E1 ubiquitin activating enzyme, the E2 ubiquitin conjugating enzymes, the E3 ubiquitin ligases, which compose the ubiquitination cascade. Given the importance of ubiquitination in several fundamental biological pathways, this process has become with no surprise the target of search and development of pharmacological drugs.
Tripartite Motif (TRIM) proteins participate in ubiquitin and ubiquitin-like modifications and form a large family of over 70 RING-type ubiquitin E3 ligases that allow proper transfer of the ubiquitin moiety to the target. Besides the N-terminal RING domain, the TRIM proteins display one or two B-box domain(s) and a Coiled-coil region responsible for their homo- and hetero-interactions. The combination of these 3 domains, the tripartite motif, is the hallmark of this class of proteins.
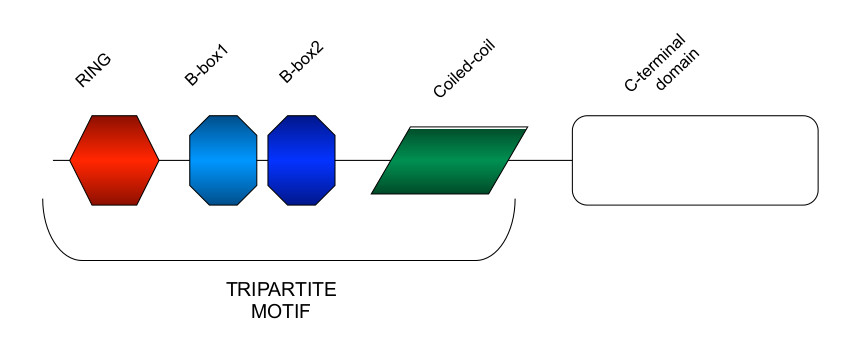
TRIM proteins are involved in many diseases and clinically relevant processes emphasizing the need for deep knowledge of TRIM proteins mechanism of action in order to design and develop targeting strategies. The ultimate goal of TRIM-NET is determining how TRIM proteins contribute to disease and how we can use this information to develop novel therapeutics for TRIM family members that are involved in common and rare pathologies, thus heavily impacting on public health.
Scientific Objectives
The TRIM-NET consortium is designed to bring together a multidisciplinary team that connects different expertise (genetics, biochemistry, molecular and cell biology, chemical biology, structural biology) across Europe to address TRIM proteins activity and to develop novel therapeutics for the treatment of different types of cancer, rare genetics diseases, neuromuscular pathologies, and neurodegenerative diseases. The TRIM-NET training programme aims at: i) defining TRIM patho-physiological functions in different disease areas; ii) developing strategies to modulate TRIM proteins activity; iii) developing high throughput screening (HTS) assays for TRIM activity; iv) validating the novel therapeutic avenues in in vitro and in vivo disease models.
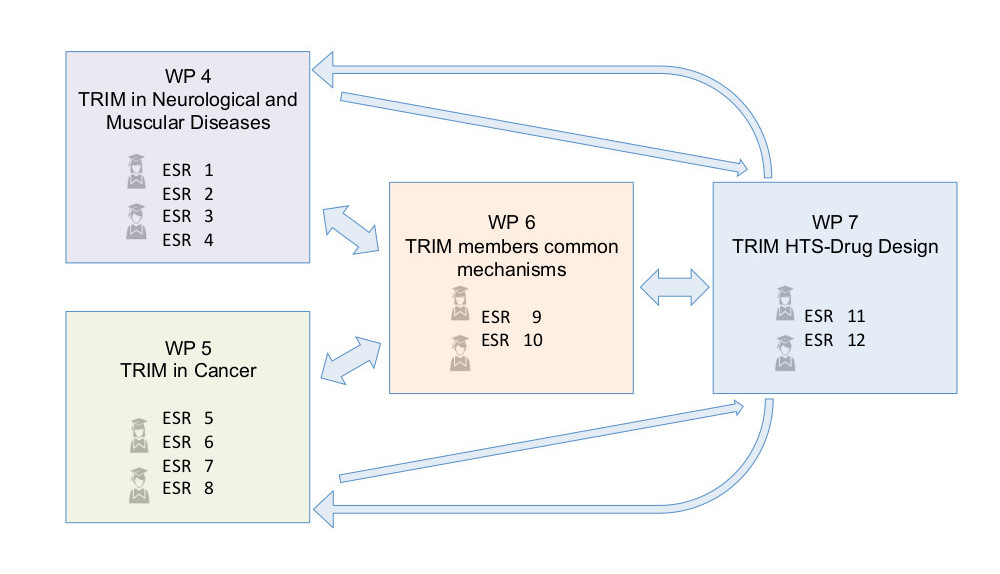
These objectives are interconnected and implemented through 4 scientific work packages (WPs):
- Several TRIM Ubiquitin E3 ligases are implicated in congenital or late-onset disorders, often though, they exert pleiotropic functions due to their multiple target specificity. It is thus crucial to define TRIM disease-specific mechanisms for the design of appropriate therapeutic strategies. WP4 will define the pathogenetic role of selected TRIM members implicated in neurodegenerative and neuromuscular disorders and use these findings to envisage therapeutic strategies.
- WP5 will define the pathogenetic role of selected TRIM family members in different types of cancers. Deregulation of E3 ligase proteasome-mediated stability of oncogenes and tumour suppressors plays an important role in onset and progression of many cancer types. TRIM proteins are no exception and several family members are implicated in a plethora of tumour types. WP5 will finally investigate how to use these findings for drug discovery.
- WP6 will define TRIM common mechanisms of action for the identification of shared and private screening and targeting strategies. TRIM proteins shared domain structure and their role in ubiquitin and ubiquitin-like target modification support this effort towards common targeting strategies.
- The goal of the research programme is to contribute to define appropriate targeting strategies for selected TRIM proteins. WP7 will design targeting strategies, peptide and/or small molecules, developing assays for high-throughput screening and performing compound library screens for TRIM proteins under study within the consortium.
The research goals above will form the basis of the research training programme for the ESRs recruited within this network. They will carry on their own project but at the same time, through intense network-wide activities and secondments, be aware of the common goal and contribute to the complete stream from disease target to drug development initiatives with exposure to interdisciplinary and intersectorial environments.
